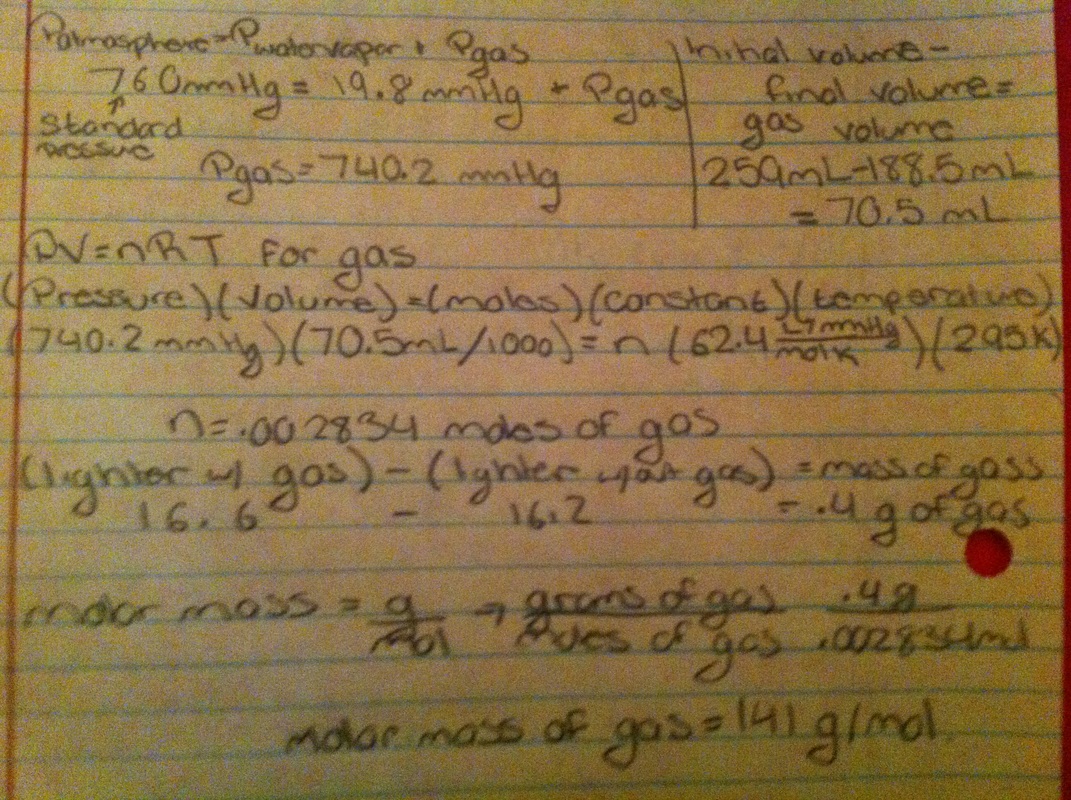Procedure:
Materials: various sizes of graduated cylinders, a plastic bin, a glass jar, a thermometer, a triple-bean balance, and a lighter.
1) The amount of water the glass jar could hold was determined by using a graduated cylinder to pour measured amounts of water in.
The water was then poured out of the glass jar.
2) The mass of the lighter was taken using a triple bean balance.
3) A plastic bin was filled with an adeqaute, about a tenth untill the brink, water and the temperature of the water was taken.
4) The vapor pressure of the water was found by applying it to the given chart in regards to temperature.
5) The glass jar, without the cap, was submerged into the plastic bin, filled with water.
6) All the air bubbles inside the jar were carefully tapped out and removed, untill it was completely filled with water.
7) The lighter was then submerged under water and inserted into the glass jar.
8) The push button was pressed and the gas inside the lighter was released into the glass jar, and gas slowly begun tajking up space inside the jar.
9) When the jar appeared to have some amount of the gas , preferably filled to the one-third, the lighter was released and the glass jar was immediately closed tightly w,ith its according top, under water.
10) The amount of water the jar now contained was calculated using graduated cylinders, and subtracted from the total amount of water the glass jar was able to hold in order to discover the volume of the gas released.
11) The lighter was then set on a paper towel to dry, and the mass of the lighter was taken after it was dry. The difference between the initial mass and the final mass of the lighter was used to determine the mass of the gas released.
12) The pressure of the gas was calculated by using the formula: Patmosphere= Pwater vapor + Pgas.
13) The temperature of the water was used as the one for the gas because both temperatures were very close together, and it was practically impossible to calculate the temperature of a gas.
14) The pressure of the gas, the volume, the temperature of the water, and the constant gas rate were all plugged into teh equation PV=nRT, which allowed for the value for the moles to be calculated.
15)The mass of the gas was then divided by the number of moles in order to determine the molar mass.
16) The plastic bin was emptied into the drain. All other supplies were rinsed out and placed on the drying rack above the sink.
1) The amount of water the glass jar could hold was determined by using a graduated cylinder to pour measured amounts of water in.
The water was then poured out of the glass jar.
2) The mass of the lighter was taken using a triple bean balance.
3) A plastic bin was filled with an adeqaute, about a tenth untill the brink, water and the temperature of the water was taken.
4) The vapor pressure of the water was found by applying it to the given chart in regards to temperature.
5) The glass jar, without the cap, was submerged into the plastic bin, filled with water.
6) All the air bubbles inside the jar were carefully tapped out and removed, untill it was completely filled with water.
7) The lighter was then submerged under water and inserted into the glass jar.
8) The push button was pressed and the gas inside the lighter was released into the glass jar, and gas slowly begun tajking up space inside the jar.
9) When the jar appeared to have some amount of the gas , preferably filled to the one-third, the lighter was released and the glass jar was immediately closed tightly w,ith its according top, under water.
10) The amount of water the jar now contained was calculated using graduated cylinders, and subtracted from the total amount of water the glass jar was able to hold in order to discover the volume of the gas released.
11) The lighter was then set on a paper towel to dry, and the mass of the lighter was taken after it was dry. The difference between the initial mass and the final mass of the lighter was used to determine the mass of the gas released.
12) The pressure of the gas was calculated by using the formula: Patmosphere= Pwater vapor + Pgas.
13) The temperature of the water was used as the one for the gas because both temperatures were very close together, and it was practically impossible to calculate the temperature of a gas.
14) The pressure of the gas, the volume, the temperature of the water, and the constant gas rate were all plugged into teh equation PV=nRT, which allowed for the value for the moles to be calculated.
15)The mass of the gas was then divided by the number of moles in order to determine the molar mass.
16) The plastic bin was emptied into the drain. All other supplies were rinsed out and placed on the drying rack above the sink.
dATA:
|
Temperature
Water 22.0°C Atmosphere 25.0°C |
Mass
Lighter with gas 16.6 g Lighter without gas 16.2 g Mass of gas . 4 g |
Volume
Water capable of fitting inside glass jar 259 mL Water without gas inside glass jar after submerssion 188.5 mL Volume of gas 70.5 mL |
Pressure
Atmosphere = Standard 1 atm = 760 mmHg Pressure of Gas 740.2 mmHg |
Calculations:
Conclusion:
This lab was created in order to determine the molar mass of butane, a substance in a lighter. The first step taken was to take the temperature of the water inside the plastic bin and using Dalton's law of partial Pressures formula, because of the water vapor on top of the liquid:
Patmosphere = Pwater vapor + Pgas :
for the atmospheric pressure, the standard pressure was plugged in which is 760 mmHg , the pressure of water was plugged into the water vapor section according to the chart that related the water temperature with its pressure. The value for the pressure of the gas was then calculated. Then the mass of the lighter before and after the gas was released from the lighter and submerged the water. The final mass was then subtracted from the initial mass in order to determine the mass of the gas that was released. The volume of the water inside the glass jar was taken without the gas and with the gas, when the lighter’s gas was inserted into the glass jar. The final volume was then subtracted from the initial in order to calculate the volume of the gas. The pressure of the gas, the volume, the temperature in Kelvin of the water( since the water’s temperature was
extremely close to the gas’), and the corresponding gas constant were plugged into the formula PV = nRT , still assuming the gas is an ideal gas, in order to figure out the moles of the gas that were used. Then the mass of the gas was divided by the number of moles in order to determine the molar mass which resulted in a value off 141 g/mol. Though, there were some sources of error: all of the gas bubbles were not removed from the glass jar when it was submerged, some of the gas from the lighter was released into the plastic bin and not into the jar( affecting the volume and the mass of the gas), and the lighter may have still had droplets of waater inside which would have affected the mass.
Patmosphere = Pwater vapor + Pgas :
for the atmospheric pressure, the standard pressure was plugged in which is 760 mmHg , the pressure of water was plugged into the water vapor section according to the chart that related the water temperature with its pressure. The value for the pressure of the gas was then calculated. Then the mass of the lighter before and after the gas was released from the lighter and submerged the water. The final mass was then subtracted from the initial mass in order to determine the mass of the gas that was released. The volume of the water inside the glass jar was taken without the gas and with the gas, when the lighter’s gas was inserted into the glass jar. The final volume was then subtracted from the initial in order to calculate the volume of the gas. The pressure of the gas, the volume, the temperature in Kelvin of the water( since the water’s temperature was
extremely close to the gas’), and the corresponding gas constant were plugged into the formula PV = nRT , still assuming the gas is an ideal gas, in order to figure out the moles of the gas that were used. Then the mass of the gas was divided by the number of moles in order to determine the molar mass which resulted in a value off 141 g/mol. Though, there were some sources of error: all of the gas bubbles were not removed from the glass jar when it was submerged, some of the gas from the lighter was released into the plastic bin and not into the jar( affecting the volume and the mass of the gas), and the lighter may have still had droplets of waater inside which would have affected the mass.
Analysis:
1) The molar mass of butane, based on the calculations, was 141g/mol.
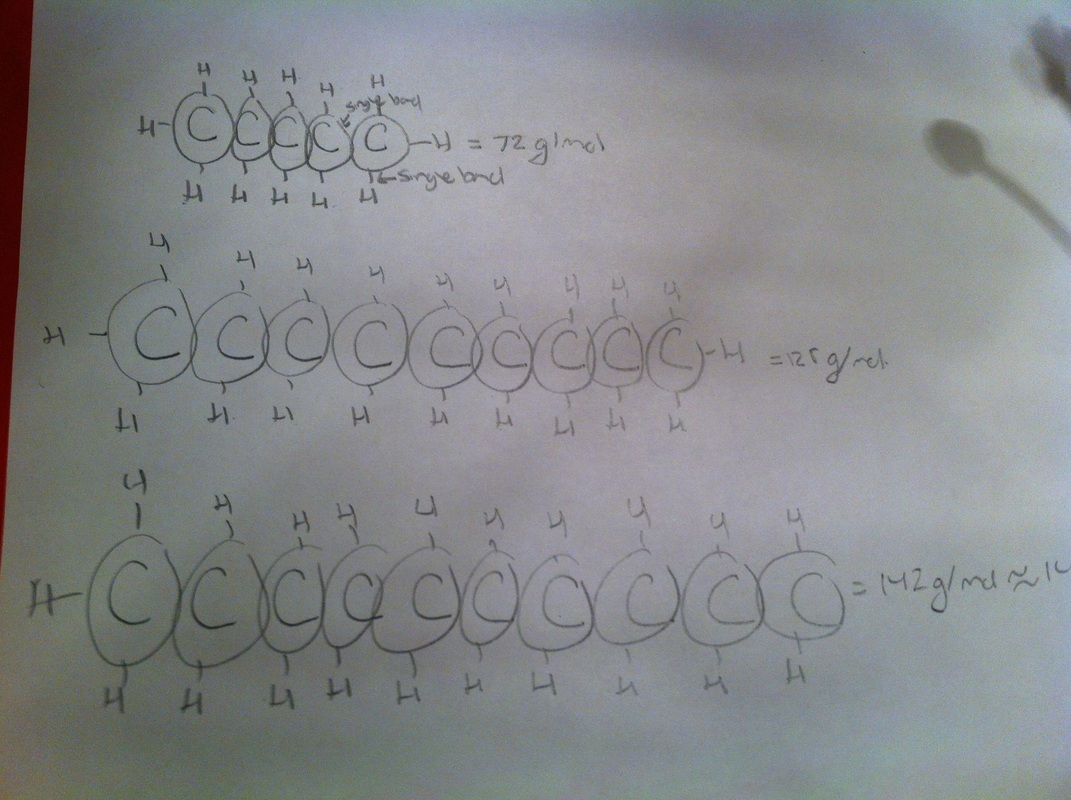
2) Based on the calculated molar mass and using the formula 141= 12c + 1h; where c represents the moles of Carbon and h the moles of Hydrogen. It was calculated that the possible formula for suggested for thegas was H22C10. Work demonstrated below.
3) Percent error Formula :
l accepted –experimental l x 100
Accepted
Accepted: C4H10 = molar mass of 58 g/mol
Experimental:C9H12 = molar mass of 141 g mol
l 58 g/mol – l41 g/mol l x 100 ]
58 g/mol
= 143% error
4)
A. If the temperature was not changed from Celsius to Kelvin the moles value in the equation PV=nRT would have been too high, which would have caused a lower molar mass for the overall gas. Because they are inversely related.
B. If the pressure was forgotten to be changed to the vapor pressure of water the mole value would not be affected greatly because
both pressures are relatively close. When rounded to the nearest tenth place both molar masses of the incorrect and the correct should be relatively close.
C. If there were air bubbles inside the gas before the lighter gas was released into the glass jar the calculated volume for the gas would have been recorded at too high a value because it also included the volume the air bubbles took up. When plugged into the equation PV=nRT it
would also produce too high a value for the variable corresponding to moles. When calculated it would also result in a molar mass way below the one that should have resulted.
D. If the lighter was not completely dry when weighed the second time, it would have demonstrated a mass value higher than it should have been because it is also including the mass of the water. A source of error, if this even occurred, might be the mass was higher than the initial. If it was not higher than the initial the value would have produced a lower mass for the mass of the gas, which would have also caused a lower molar
mass for butane because they are directly related.
5) Based on the results from this experiment the other gasses have a higher molar mass than butane. When calculated the experimental value was 141 g/mol the actual molar mass for butane is 58 g/mol, the difference between the experimental and the actual is 83 grams which means the other gasses mixed in with the butane have to have an added molar mass of 83 grams which is way above butane.Considering that most of the liquid inside the lighter is butane when average the other gases should have a higher mass in order to result in a molar mass of 141g/mol. In reality, whether or not the other gases mixed with the lighter liquid have a higher mass of butane depends on the number of other gases that were present.
6) When the substance inside the lighter was release, it was released as a gas. This was due to the fact that the substance undergoes a physical state change, in sense it was evaporating. When the trigger or push button on the lighter was pressed , the gas inside was released to the outside, it dramatically changed the pressure which caused the gas to change its physical state because of the lower pressure.
2) Based on the calculated molar mass and using the formula 141= 12c + 1h; where c represents the moles of Carbon and h the moles of Hydrogen. It was calculated that the possible formula for suggested for thegas was H22C10. Work demonstrated below.
3) Percent error Formula :
l accepted –experimental l x 100
Accepted
Accepted: C4H10 = molar mass of 58 g/mol
Experimental:C9H12 = molar mass of 141 g mol
l 58 g/mol – l41 g/mol l x 100 ]
58 g/mol
= 143% error
4)
A. If the temperature was not changed from Celsius to Kelvin the moles value in the equation PV=nRT would have been too high, which would have caused a lower molar mass for the overall gas. Because they are inversely related.
B. If the pressure was forgotten to be changed to the vapor pressure of water the mole value would not be affected greatly because
both pressures are relatively close. When rounded to the nearest tenth place both molar masses of the incorrect and the correct should be relatively close.
C. If there were air bubbles inside the gas before the lighter gas was released into the glass jar the calculated volume for the gas would have been recorded at too high a value because it also included the volume the air bubbles took up. When plugged into the equation PV=nRT it
would also produce too high a value for the variable corresponding to moles. When calculated it would also result in a molar mass way below the one that should have resulted.
D. If the lighter was not completely dry when weighed the second time, it would have demonstrated a mass value higher than it should have been because it is also including the mass of the water. A source of error, if this even occurred, might be the mass was higher than the initial. If it was not higher than the initial the value would have produced a lower mass for the mass of the gas, which would have also caused a lower molar
mass for butane because they are directly related.
5) Based on the results from this experiment the other gasses have a higher molar mass than butane. When calculated the experimental value was 141 g/mol the actual molar mass for butane is 58 g/mol, the difference between the experimental and the actual is 83 grams which means the other gasses mixed in with the butane have to have an added molar mass of 83 grams which is way above butane.Considering that most of the liquid inside the lighter is butane when average the other gases should have a higher mass in order to result in a molar mass of 141g/mol. In reality, whether or not the other gases mixed with the lighter liquid have a higher mass of butane depends on the number of other gases that were present.
6) When the substance inside the lighter was release, it was released as a gas. This was due to the fact that the substance undergoes a physical state change, in sense it was evaporating. When the trigger or push button on the lighter was pressed , the gas inside was released to the outside, it dramatically changed the pressure which caused the gas to change its physical state because of the lower pressure.