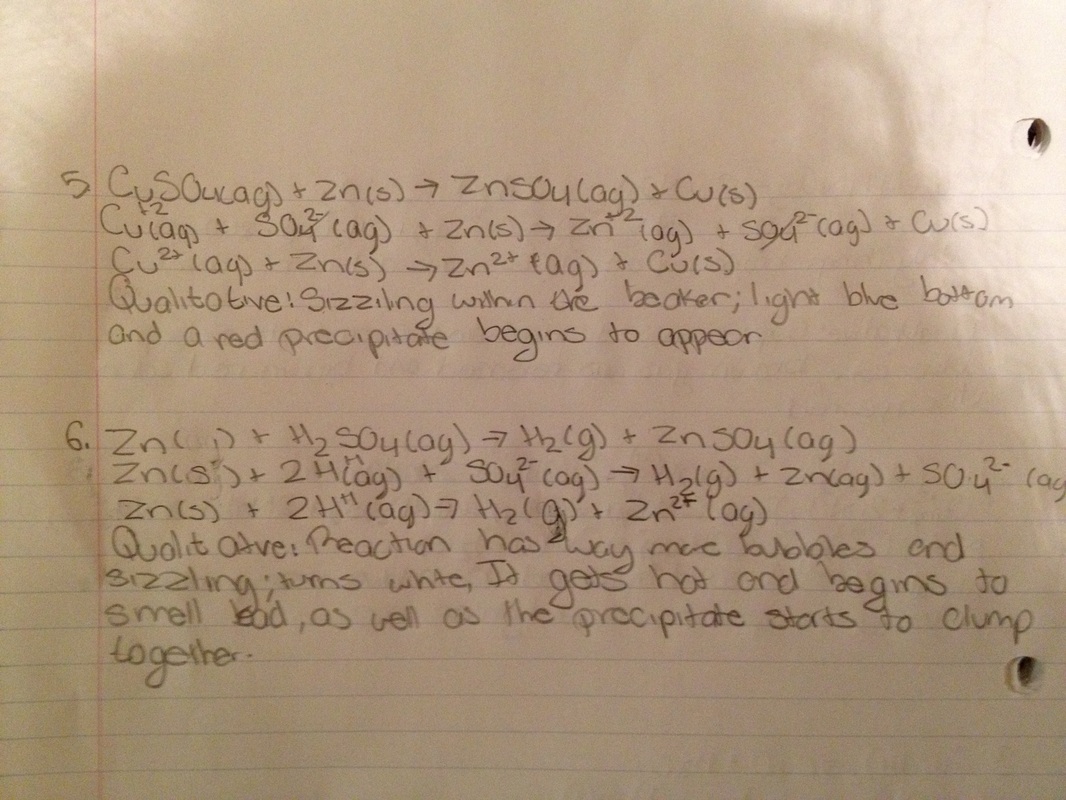Qualitative and quantitative data:
conclusions:
This lab proved the idea that copper could change from one chemical state to another, and in the end be returned to the elemental copper. In the lab, the copper was dissolved in nitrate acid which released a brown smoke and the liquid turned a pure blue. Then, the beaker was put in an ice bath and added sodium hydroxide in order to change the state to a solid. It was then headed to separate the solid from the liquid. It was decanted to get rid of the liquid. Then sulfate acid and zinc were added to get rid of the blue color and then decanted in order to get the copper. Finally it was set to dry to get rid of all the remaining liquid. The beginning mass for the copper was 2.03 grams and the resulting mass was 13.103 grams which means there was a source off error within the experiment, possible explanations and/or causes are detailed below.
Discussion of Theory:
This lab procedure was created to verify the law of conservation of mass; mass is neither created nor destroyed. The idea is that the entire mass of a material is not affected by chemical changes. The mass of the starting amount of copper should be equal to the ending mass of copper. Various chemical reactions took place and in the ending result the copper was recovered. This is due to the fact that a continuous number of molecules for copper were present during the experiment. Each step contained a specific number of moles that were present in each reaction either as a solid or a liquid. The atomic theory predicts that the starting amount of copper remains constant throughout an experiment and till the end.
Various types of reactions occurred throughout this experiment. The first one that occurred was a metallic
hydrogen nitrate in which the products were the ionic compound, water and nitrite. The copper became aqueous and dissolved in the liquid and water vapor and nitrite gas were released. The second reaction that occurred was a double
replacement reaction, in which both ionic compounds switch anions and/or cations and the products were copper (II) hydroxide and sodium nitrate. In this reaction the sodium nitrate is an aqueous compound and the copper (II) hydroxide is the insoluble product; unable to dissolve in water therefore it is the precipitate.When things are insoluble it means water molecules are unable to separate the compound into ions and surround the totality of ions. The third was a metallic hydroxide in which the results were copper oxide and water. The copper hydroxide remains a solid and is surrounded by water molecules. The forth one was a double replacement reaction in which one of the products was water and the other was copper (II) sulfate. The fifth one was a single replacement reaction involving copper and zinc; zinc is higher on the activity series than copper therefore copper is replaced with zinc and the reaction is successful. The zinc reaction is also a single replacement in which the zinc replaced the hydrogen in the sulfate acid because zinc is higher in the activity series than hydrogen. The reaction is also successful.At his point the copper has been retrived and at the end all the compounds and elements should be decanted or evaporated and the only thing left should be the solid copper. Stoichiometry problems were used in each reaction in order to balance equations. These problems were also used to calculate the amount of product which could have resulted from the amount of reactants used based on the mole to mole ration for every reaction.
During this experiment many toxic materials were used including toxic gasses nad liquid that should be prevented from inhalation and touch to the skin.Many acids are concentrated and could have caused severe burns. For acid spills, acids needed to be neutarlized before being disposed off. Safety precautions included eyeware, long clothing, and protective aprons.
The percent yield in this experiment; percentage of accuracy, being the actual product value over the expected value times one hundred. This value not reasonable in the experiment, the percent yield ended up being 645%, because they ending mass of copper was extremely massive when compare to the value that should have resulted; therefore, the experiment was not successful. Reasons for this valued are detailed below.
Various types of reactions occurred throughout this experiment. The first one that occurred was a metallic
hydrogen nitrate in which the products were the ionic compound, water and nitrite. The copper became aqueous and dissolved in the liquid and water vapor and nitrite gas were released. The second reaction that occurred was a double
replacement reaction, in which both ionic compounds switch anions and/or cations and the products were copper (II) hydroxide and sodium nitrate. In this reaction the sodium nitrate is an aqueous compound and the copper (II) hydroxide is the insoluble product; unable to dissolve in water therefore it is the precipitate.When things are insoluble it means water molecules are unable to separate the compound into ions and surround the totality of ions. The third was a metallic hydroxide in which the results were copper oxide and water. The copper hydroxide remains a solid and is surrounded by water molecules. The forth one was a double replacement reaction in which one of the products was water and the other was copper (II) sulfate. The fifth one was a single replacement reaction involving copper and zinc; zinc is higher on the activity series than copper therefore copper is replaced with zinc and the reaction is successful. The zinc reaction is also a single replacement in which the zinc replaced the hydrogen in the sulfate acid because zinc is higher in the activity series than hydrogen. The reaction is also successful.At his point the copper has been retrived and at the end all the compounds and elements should be decanted or evaporated and the only thing left should be the solid copper. Stoichiometry problems were used in each reaction in order to balance equations. These problems were also used to calculate the amount of product which could have resulted from the amount of reactants used based on the mole to mole ration for every reaction.
During this experiment many toxic materials were used including toxic gasses nad liquid that should be prevented from inhalation and touch to the skin.Many acids are concentrated and could have caused severe burns. For acid spills, acids needed to be neutarlized before being disposed off. Safety precautions included eyeware, long clothing, and protective aprons.
The percent yield in this experiment; percentage of accuracy, being the actual product value over the expected value times one hundred. This value not reasonable in the experiment, the percent yield ended up being 645%, because they ending mass of copper was extremely massive when compare to the value that should have resulted; therefore, the experiment was not successful. Reasons for this valued are detailed below.
Sorces of error:
The yield from copper was extremely high; the final mass was greater than the initial mass of copper- a difference greater than ten. The previous could be the result of a large quantity of zinc still being present in the evaporating dish when the copper was measured. Possibly, the zinc did not fully react with sulfuric acid, and most of it remained in its natural form because too much zinc was added, to little sulfate acid, or the reaction was not allowed to be completely finished . Also, maybe the copper was not entirely dry when measured and the volume of water ,still in the copper ,affected the overall mass when the copper was weighed. This was not expected, if anything, a lower yield was expected when compared to the initial mass of copper. Traces of material were left on the instruments used, such as the beaker and the stirring rod. as well as material stuck to the walls of the beaker that were extremley difficult to remove. Also, there is a possibility that material was also lost when decanting was performed, either from unreacted copper or the overall product.
questions:
1) The product of the reaction between copper and nitric acid was placed on ice in order to solidify its form, freeze it.
2) In step four the reaction that occurred was Cu(NO3)2 + 2NaOH → Cu(OH)2 + 2NaNO3 , which was a double-replacement reaction since both were ionic compounds and the copper traded its nitrate ion for the hydroxide ion. During this reaction a precipitate was also formed, the copper hydroxide. In step seven the reaction that occurred was CuO + H2SO4 → CuSO4 + H2O, which was also a double replacement reaction in which one of the products was water and the solid Copper (II) sulfate. In step nine the reaction that occurred was Zn + H2SO4 → H2 + ZnSO4, whch was a single replacement reaction in which the zinc replaced the hydrogen in the sulfate acid because zinc is higher in the activity series than hydrogen.
3) The reaction of excess zinc with sulfuric acid is, Zn + H2SO4 → H2 + ZnSO4. If this reaction did not fully react, or was an incomplete reaction , the zinc did not react with the sulfate acid and remained with copper, and the solid would consist of copper and zinc. When measured, the mass would contain both the mass of the copper and the mass of the zinc, in which case, giving an inaccurate mass of copper way above the initial.
4) The reaction when copper oxide was cleaned was CuO + H2SO4 → CuSO4 + H2O, the net ionic equation , CuO(S) + H2+1 (aq) + SO4 -2(aq) →Cu +2(aq) + SO4-2(aq)+ H20(l), the ion that can be removed from this equation is sulfate acid, H2SO4 -2. The resulting net ionic equation is CuO + H2→ Cu + H2O, the sulfate acid was decanted.
5) During the first time sulfate acid was added the copper was in an aqueous state, when the sulfate acid was added again to the solution the state of matter for copper was a solid.
6) When the copper precipitate was washed the reaction that occurred was Zn + H2SO4 → H2 + ZnSO4: Zn(aq) + H2 (aq)+1 + SO4(aq’0 -2 → H2(aq) +1 + Zn(aq) +2 + SO4(aq) -2 , all are ions therefore when washed the only thing that should remain is the copper precipitate.
2) In step four the reaction that occurred was Cu(NO3)2 + 2NaOH → Cu(OH)2 + 2NaNO3 , which was a double-replacement reaction since both were ionic compounds and the copper traded its nitrate ion for the hydroxide ion. During this reaction a precipitate was also formed, the copper hydroxide. In step seven the reaction that occurred was CuO + H2SO4 → CuSO4 + H2O, which was also a double replacement reaction in which one of the products was water and the solid Copper (II) sulfate. In step nine the reaction that occurred was Zn + H2SO4 → H2 + ZnSO4, whch was a single replacement reaction in which the zinc replaced the hydrogen in the sulfate acid because zinc is higher in the activity series than hydrogen.
3) The reaction of excess zinc with sulfuric acid is, Zn + H2SO4 → H2 + ZnSO4. If this reaction did not fully react, or was an incomplete reaction , the zinc did not react with the sulfate acid and remained with copper, and the solid would consist of copper and zinc. When measured, the mass would contain both the mass of the copper and the mass of the zinc, in which case, giving an inaccurate mass of copper way above the initial.
4) The reaction when copper oxide was cleaned was CuO + H2SO4 → CuSO4 + H2O, the net ionic equation , CuO(S) + H2+1 (aq) + SO4 -2(aq) →Cu +2(aq) + SO4-2(aq)+ H20(l), the ion that can be removed from this equation is sulfate acid, H2SO4 -2. The resulting net ionic equation is CuO + H2→ Cu + H2O, the sulfate acid was decanted.
5) During the first time sulfate acid was added the copper was in an aqueous state, when the sulfate acid was added again to the solution the state of matter for copper was a solid.
6) When the copper precipitate was washed the reaction that occurred was Zn + H2SO4 → H2 + ZnSO4: Zn(aq) + H2 (aq)+1 + SO4(aq’0 -2 → H2(aq) +1 + Zn(aq) +2 + SO4(aq) -2 , all are ions therefore when washed the only thing that should remain is the copper precipitate.